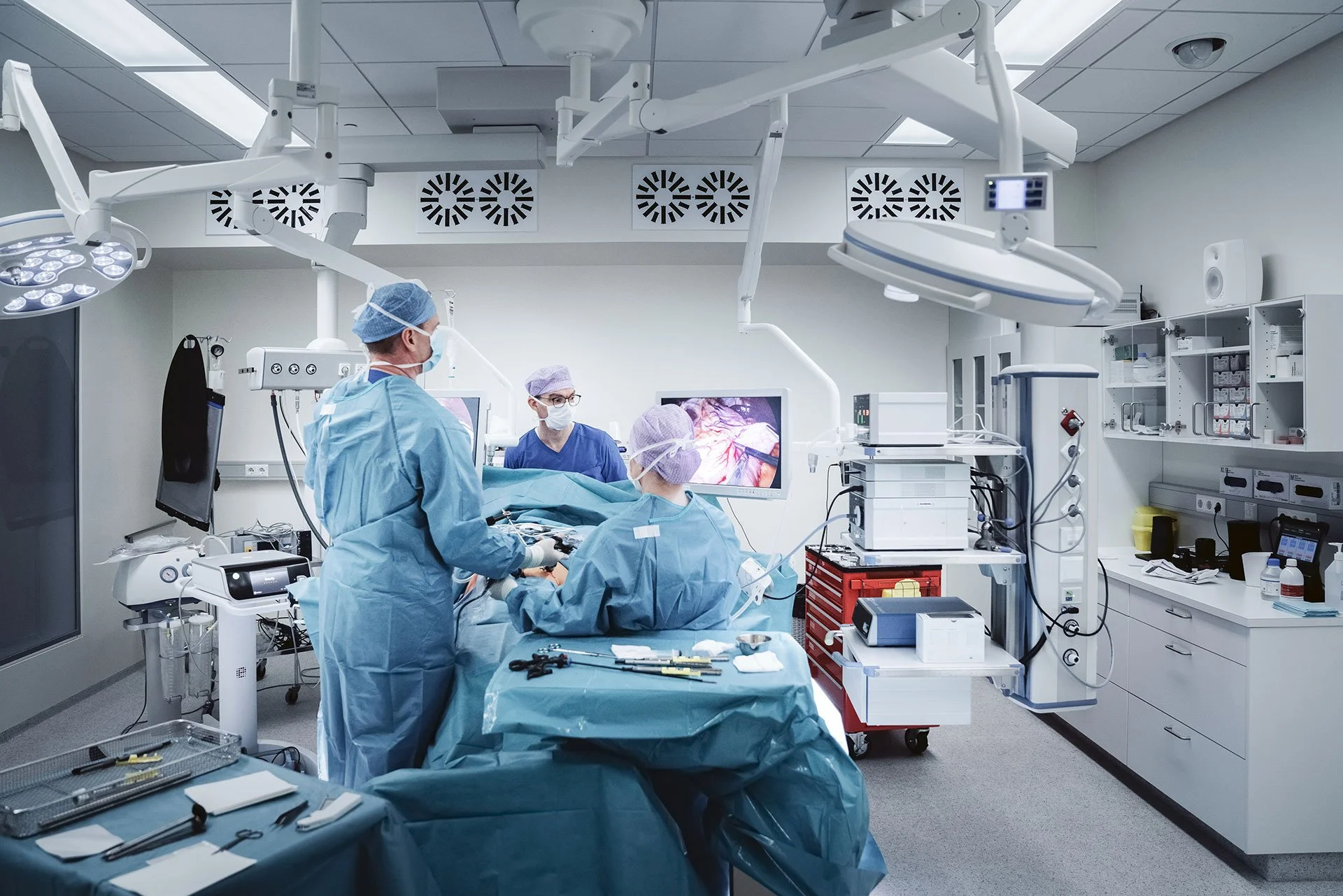
About us
At Mariner Endosurgery, our mission is to design and develop medical devices that support surgical safety programs for laparoscopic and minimally invasive surgery (MIS) procedures.
The company was founded, and several technology patents were granted as a part of a robust IP portfolio. This would become the company’s flagship, niche surgical safety solution.
Company History
The inception of the Endosurgery business occurred, focused on improving surgical safety in minimally invasive procedures.
A milestone achieved; Mariner received 510K FDA Clearance on its LaparoGuard™ Surgical Solution with Safe Zone definition.
To support pandemic supply efforts Mariner engaged with federal and provincial health systems for the distribution of medical and surgical supplies.
The organization continues to invest in its innovation and development pipeline, including a redeveloped product with LaparoGuard™ AR.
See for yourself
Let us show you how easy it is to integrate Mariner Endosurgery’s innovative safety technology into your surgical practice today.





